 Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. 2 Nicely accounted by the Oxford Dictionary when referring to the periodic table as: ‘A table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure (and hence similar chemical properties) appear in vertical columns’. As an alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, its angular momentum, and an angular momentum vector component ( magnetic quantum number). 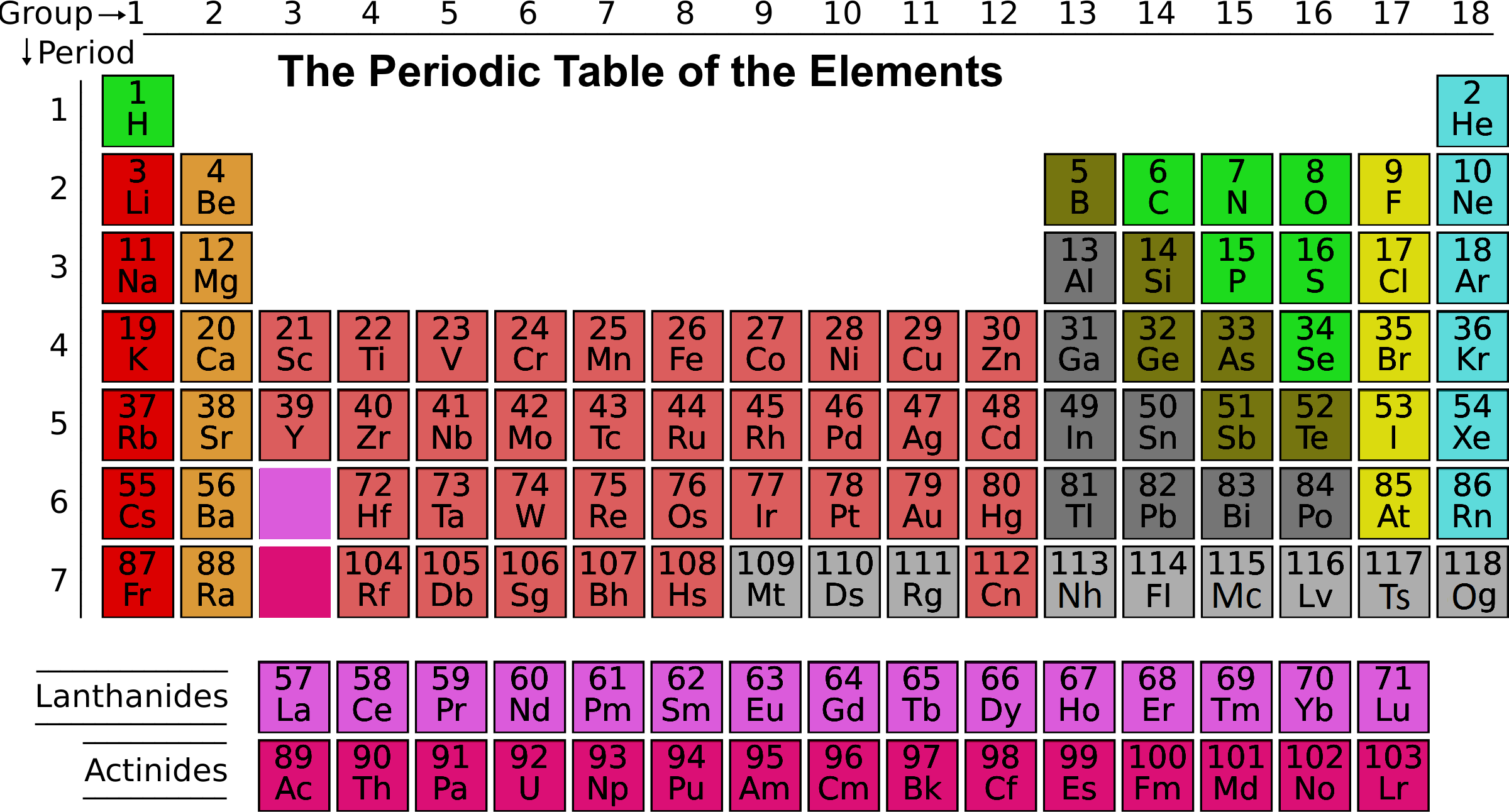
The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. 
This is because the outermost electron is, on average, farther from the nucleus, meaning it is held less. On the periodic table, first ionization energy generally decreases as you move down a group. In atomic theory and quantum mechanics, an atomic orbital ( / ˈ ɔːr b ɪ t ə l/) is a function describing the location and wave-like behavior of an electron in an atom. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. An elements first ionization energy is the energy required to remove the outermost, or least bound, electron from a neutral atom of the element. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depends on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. For the collection of spaceflight orbits, see Orbital shell (spaceflight). These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity."Orbital shell" redirects here. This happens because the number of filled principal energy levels (which shield the outermost electrons from attraction to the nucleus) increases downward within each group. Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. First, electrons are added one at a time moving from left to right across a period. 
In addition to this activity, there are two other important trends. Stable octets are seen in the inert gases, or noble gases, of Group VIII of the periodic table. Elements tend to gain or lose valence electrons to achieve stable octet formation. /PeriodicTableWallpaper-56a12a3a3df78cf772680422.jpg)
These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. An interactive Periodic table can be found Periodic Table of the Elements, LibreTexts. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
